6-APDB
| Summary sheet: 6-APDB |
| 6-APDB | |||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Chemical Nomenclature | |||||||||||||||||||||||||||||||
| Common names | 6-APDB | ||||||||||||||||||||||||||||||
| Substitutive name | 6-(2-aminopropyl)-2,3-dihydrobenzofuran | ||||||||||||||||||||||||||||||
| Systematic name | 1-(2,3-dihydrobenzofuran-6-yl)propan-2-amine | ||||||||||||||||||||||||||||||
| Class Membership | |||||||||||||||||||||||||||||||
| Psychoactive class | Entactogen / Stimulant | ||||||||||||||||||||||||||||||
| Chemical class | Amphetamine / Benzofuran | ||||||||||||||||||||||||||||||
| Routes of Administration | |||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
| Interactions | |||||||||||||||||||||||||||||||
| Alcohol | |||||||||||||||||||||||||||||||
| MXE | |||||||||||||||||||||||||||||||
| Dissociatives | |||||||||||||||||||||||||||||||
| DXM | |||||||||||||||||||||||||||||||
| MDMA | |||||||||||||||||||||||||||||||
| Stimulants | |||||||||||||||||||||||||||||||
| 25x-NBOMe | |||||||||||||||||||||||||||||||
| 25x-NBOH | |||||||||||||||||||||||||||||||
| Tramadol | |||||||||||||||||||||||||||||||
| MAOIs | |||||||||||||||||||||||||||||||
| SNRIs | |||||||||||||||||||||||||||||||
| Serotonin releasers | |||||||||||||||||||||||||||||||
| SSRIs | |||||||||||||||||||||||||||||||
| 5-HTP | |||||||||||||||||||||||||||||||
6-APDB (also known as 6-(2-aminopropyl)-2,3-dihydrobenzofuran or 4-Desoxy-MDA) is a stimulant and entactogenic research chemical of the phenethylamine and benzofuran classes. It is a closely related synthetic analogue of MDA and 6-APB and broadly shares the characteristics of serotonin-selective triple monoamine releasers and reuptake inhibitors associated with other entactogenic or empathogenic compounds.
6-APDB was first synthesized and studied along with 5-APDB in 1993 by David E. Nichols as a potential non-neurotoxic alternative to MDMA[1]. It did not come into popular recreational use until over a decade later, where it briefly entered the rave scene and global research chemicals market, in particular the "legal highs" market in the U.K., before its sale and import were subsequently banned.
Because 6-APDB and other substituted benzofurans have not been explicitly outlawed in some countries, they are often technically legal, contributing to their popularity as a substitute or replacement for serotonergic entactogens like MDMA or MDA, and are typically distributed through the online research chemicals grey market.
Chemistry
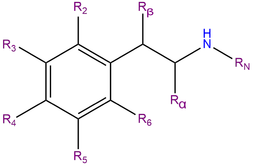
6-APDB, also known as 6-(2-aminopropyl)-2,3-dihydrobenzofuran, is a synthetic molecule of the benzofuran family. Molecules of this class contain a phenethylamine core bound to an amino (NH2) group through an ethyl chain with an additional methyl substitution at Rα.
6-APDB does not contain a methyl substitution on RN, a motif which it shares with MDA. It is an analogue of MDA where the heterocyclic 4-position oxygen from the 3,4-methylenedioxy ring has been replaced with a methylene bridge. 6-APDB shares this furan ring with 5-APDB, 5-MAPDB and 6-MAPDB.
Pharmacology
6-APDB acts as a releasing agent and triple reuptake inhibitor of the monoamine neurotransmitters known as serotonin, dopamine and noradrenaline[2] which are the global neurotransmitters that modulate the brain's ability to feel pleasure, motivation, reward, planning, attention and focus. This is done by promoting the release and inhibiting the reuptake and reabsorption of the neurotransmitters after they have performed their function of transmitting a neural impulse through release into the synaptic cleft, essentially allowing them to accumulate and render them liable for immediate reuse. The net result is excitation in a manner which causes a combination of physically stimulating, relaxing, disinhibiting and euphoric effects.[3]
The unsaturated benzofuran derivative 6-APB, or 6-(2-aminopropyl)benzofuran is also known, but the difference in pharmacological effects between 6-APB and 6-APDB has yet to be fully elucidated.
Subjective effects
Disclaimer: The effects listed below cite the Subjective Effect Index (SEI), an open research literature based on anecdotal user reports and the personal analyses of PsychonautWiki contributors. As a result, they should be viewed with a healthy degree of skepticism.
It is also worth noting that these effects will not necessarily occur in a predictable or reliable manner, although higher doses are more liable to induce the full spectrum of effects. Likewise, adverse effects become increasingly likely with higher doses and may include addiction, severe injury, or death ☠.
Physical effects 
-
- Spontaneous physical sensations - The "body high" of 6-APDB can be described as a moderate to extreme euphoric tingling sensation that radiates throughout the entire body. It is capable of becoming overwhelmingly pleasurable at higher doses, and capable of immobilizing the user. This sensation maintains a consistent presence that steadily rises with the onset and hits its limit once the peak has been reached.
- Stimulation and Sedation - In terms of its effects on the user's physical energy levels, 6-APDB is commonly considered to have the paradoxical ability to both be stimulating as well as sedating and relaxing. Overall, it is thought to be far less energetic than MDMA or MDA and tends to exert more of a pronounced sedating stoning or couch-locking effect. Unlike MDMA, this does not particularly encourage activities such as running, climbing and dancing in a way that makes MDMA a popular choice for musical events such as festivals and raves. The particular style of stimulation which 6-APDB presents is far less forceful in a way that is more reminiscent of mescaline.
- Vibrating vision - At high doses, a person's eyeballs may begin to spontaneously wiggle back and forth in a rapid motion, causing the vision to become blurry and temporarily out of focus. This is a condition known as nystagmus.
- Dehydration - Feelings of dry mouth and dehydration are a universal experience with this class of compounds; this effect is a product of an increased heart rate and bodily metabolism. While it is important to avoid becoming dehydrated (especially when out dancing in a hot environment) there have been a number of notable cases of users suffering from water intoxication through over-drinking (in order to compensate). So it is advised that users simply sip at water and avoid over-drinking.
- Difficulty urinating - Like with other triple releasers, higher doses of 6-APDB result in an overall difficulty when it comes to urination. This is an effect that is completely temporary and harmless. It is due to 6-APDB’s promotion of the release of anti-diuretic hormone (ADH); ADH is responsible for regulating urination. This effect can be lessened by simply relaxing, but can be significantly relieved by placing a hot flannel over the genitals to warm them up and encourage blood flow to the region.
- Appetite suppression
- Bodily control enhancement
- Increased blood pressure
- Increased bodily temperature - As 6-APDB is a serotonin releasing agent, a rise in core body and brain temperature tends to be high and consistent throughout the experience. Caution must be taken as too high of a dose can result in the dysregulation of the brain's ability to regulate its internal core temperature, which can result in serotonin syndrome, a condition which can be fatal if left untreated.
- Increased heart rate
- Increased perspiration
- Perception of bodily lightness
- Physical euphoria
- Pupil dilation
- Stamina enhancement
- Tactile enhancement
- Teeth grinding - This is usually only present at moderate to higher doses and is similar to what one might experience from MDMA or MDA.
- Temporary erectile dysfunction
Visual effects 
-
Similar to MDMA, the visual effects of 6-APDB have an occurrence rating that is more selective and less consistent than any of the traditional psychedelics. This is to the point where many people disregard psychedelic experiences within 6-APDB as a "myth" or "rumour", but this is simply because they have not experienced it for themselves. The effects can never be guaranteed to manifest themselves, but are more likely to occur with chemically pure, high dose 6-APDB experiences, towards the end of the experience and if the user has been smoking cannabis. They are also more likely to occur if the user has prior experience with psychedelics, but also remain entirely possible within those who have never tried one them as well.
Unlike MDMA, 6-APDB has the capacity to directly induce mild to moderate visual effects (due to its partial agonism of the 5HT2A receptor), which makes it qualitatively more comparable to MDA.
Enhancements
6-APDB presents an array of visual enhancements which are mild in comparison to traditional psychedelics, but still distinctively present. These generally include:
Distortions
Geometry
The visual geometry that is present throughout this trip can be described as more similar in appearance to that of psilocin than LSD. It can be comprehensively described through its variations as primarily intricate in complexity, abstract in form, organic in style, structured in organization, dimly lit in lighting, mostly monotone in colour with blues and greys, glossy in shading, sharp in edges, small in size, fast in speed, smooth in motion, equal in round and angular corners, non-immersive in depth and consistent in intensity. At higher doses, they are significantly more likely to result in states of level 8A visual geometry over level 8B.
Hallucinatory states
6-APDB is capable of producing a unique range of low and high level hallucinatory states in a fashion that is significantly less consistent and reproducible than that of many other commonly used psychedelics. These effects are far more common during the offset of the experience and commonly include:
- External hallucination (autonomous entities; settings, sceneries, and landscapes; perspective hallucinations and scenarios and plots) - This effect is very similar to the same experience found within deliriants, but does not manifest itself consistently and usually happens only at high doses. It can be comprehensively described through its variations as delirious in believability, autonomous in controllability and solid in style. They usually follow themes of memory replays and semi-realistic or expected events. For example, people could be casually holding objects or performing actions which one would expect them to be in real life before disappearing and dissolving under further inspection. Common examples of this include seeing people wearing glasses or hats when they are not and mistaking faces of your friends for random people, and objects as human beings or animals.
- Internal hallucination - The internal hallucinations which 6-APDB induces are generally only present as spontaneous breakthroughs at extremely high doses. This effect's variations are delirious in believability, interactive in style, new experiences in content, autonomous in controllability and solid in appearance. The most common way in which they manifest themselves are through hypnagogic scenarios which the user may experience as they are drifting off to sleep after a night of use; these can usually be described as memory replay from the previous several hours. These are short and fleeting, but frequent and completely believable and convincing as they happen. In terms of the theme, they often take the form of conversations with the people who were with you or instead manifest themselves as bizarre and extremely nonsensical plots.
- Peripheral information misinterpretation
Cognitive effects 
-
The cognitive effects of 6-APDB can be broken down into several components which progressively intensify proportional to dosage. The general head space of 6-APDB is described by many as one of moderate mental stimulation, feelings of love, openness or empathy, and powerful euphoria. It displays a large number of typical psychedelic, entactogenic and stimulant cognitive effects.
The most prominent of these cognitive effects generally include:
- Cognitive euphoria - Strong emotional euphoria and feelings of happiness are present within 6-APDB and are likely a direct result of serotonin and dopamine release.
- Empathy, love, and sociability enhancement - This particular effect is generally more consistent, pronounced, powerful and therapeutic with 6-APDB than any other known substance. It is the most obvious and noticeable effect within any 6-APDB experience and dominates the head space. With time, repeated use, and improper spacing, however, this effect becomes severely diminished as the perspective it instills becomes fully imprinted, making it so users feel merely speedy and scattered with no new found urges to communicate or bond with others.
- Time distortion - Strong feelings of time compression are common within 6-APDB and alter the experience of time quite noticeably.
- Unity and interconnectedness - Experiences of unity, oneness and interconnectedness between level 2 - 3 are common within 6-APDB. This component most consistently manifests itself at high doses within large crowds at raves and musical events in the form of "becoming one with the crowd."
- Anxiety suppression
- Compulsive redosing - Due to its potential euphoria-inducing effects, there is the potential for 6-APDB to encourage compulsive redosing, much like with MDMA or MDA. Yet due to the length of the experience, many find this less of an issue.
- Creativity enhancement
- Existential self-realization - Although this effect is present, it is not quite as pronounced or as consistent when compared to other hallucinogens such as mescaline, LSD or MXE. Due to the relative calmness and lack of chaotic energy that 6-APDB possesses relative to MDMA, however, this combined with its extended duration may make it a better therapeutic agent and can be thought of as lying closer to the spectrum of mescaline than MDMA.
- Focus enhancement - This component is most effective at low to moderate doses as anything higher will usually impair concentration.
- Immersion enhancement
- Increased libido
- Increased music appreciation
- Mindfulness
- Motivation enhancement
- Thought acceleration
- Wakefulness - This component is present, but to a noticeably lesser degree than MDMA. Users often report being heavily sedated or "floored" compared to typical stimulants.
Auditory effects 
After effects 
-
The effects which occur during the offset of a stimulant experience generally feel negative and uncomfortable in comparison to the effects which occurred during its peak. This is often referred to as a "comedown" and occurs because of neurotransmitter depletion. Its effects commonly include:
- Anxiety
- Appetite suppression
- Brain zaps - This effect is typically seen following large or repeated dosing and is thought to be associated with disruptions to the serotonin system (as they are also observed with the sudden discontinuation of SSRIs).
- Cognitive fatigue
- Depression
- Dream potentiation - Some users note extremely strange and sometimes scary dreams for several nights after taking large doses of 6-APDB (a feature it shares with other strong serotonin-releasing agents).
- Irritability
- Motivation suppression
- Thought deceleration
- Wakefulness
Experience reports
There are currently no anecdotal reports which describe the effects of this compound within our experience index. Additional experience reports can be found here:
Toxicity and harm potential
Due to only having a short history of human use, the toxicity and harm potential is not exactly known. Due to its similarity to MDMA, it is likely that the administration of repeated or high dosages of 6-APDB can be neurotoxic and cardiotoxic[4][5] in some form.
The exact toxic dosage is unknown. It is strongly recommended that one use harm reduction practices when using this drug.
Short-term health concerns
Short-term physical health risks of 6-APDB consumption include dehydration, insomnia, and hyperthermia.[6] Continuous activity without sufficient rest or rehydration may cause body temperature to rise to dangerous levels, and loss of fluid via excessive perspiration puts the body at further risk as the stimulatory and euphoric qualities of the drug may render the user oblivious to their energy expenditure for quite some time. Diuretics such as alcohol may exacerbate these risks further, although this is known to be more of a problem for MDMA than it is 6-APDB.
Although it has not been formally studied, like with MDMA, small changes in ambient temperature may cause large changes in 6-APDB-induced serotonin neurotoxicity and core body temperature in the rat.[7][8]
Long-term health concerns
The neurotoxicity of 6-APDB is controversial. It was specifically designed to be less neurotoxic than MDA or MDMA by circumventing the production of certain metabolic byproducts thought to underlie their toxicity (specifically alpha-methyl-dopamine).[citation needed] Although it is likely to be physically safe to try in a responsible context, it is completely possible that the administration of repeated or high dosages of 6-APDB could result in neurotoxic effects in some form, possibly manifesting as deficits in cognitive, affective and psychomotor function.
As with MDMA, long-term heavy use of 6-APDB is likely cardiotoxic and thought to lead to valvulopathy through its actions on the 5-HT2B receptor.[4][5]
Tolerance and addiction potential
As with other stimulants, the chronic use of 6-APDB can be considered moderately addictive with a high potential for abuse and is capable of causing psychological dependence among certain users. When addiction has developed, cravings and withdrawal effects may occur if a person suddenly stops their usage.
As a potent releaser of serotonin, tolerance builds quickly with prolonged and repeated use to the point that the drug eventually loses any positive effects and instead leaves the user in an uncomfortable state of anxious stimulation and dysphoria. This results in users having to administer increasingly large doses to achieve the same effects. After that, it takes about 21-30 days for the tolerance to be reduced to half and 2-3 months to be back at baseline (in the absence of further consumption). 6-APDB presents cross-tolerance with all dopaminergic stimulants, meaning that after the consumption of 6-APDB all stimulants will have a reduced effect.
Dangerous interactions
Warning: Many psychoactive substances that are reasonably safe to use on their own can suddenly become dangerous and even life-threatening when combined with certain other substances. The following list provides some known dangerous interactions (although it is not guaranteed to include all of them).
Always conduct independent research (e.g. Google, DuckDuckGo, PubMed) to ensure that a combination of two or more substances is safe to consume. Some of the listed interactions have been sourced from TripSit.
- 25x-NBOMe & 25x-NBOH - 25x compounds are highly stimulating and physically straining. Combinations with 6-APDB should be strictly avoided due to the risk of excessive stimulation and heart strain. This can result in increased blood pressure, vasoconstriction, panic attacks, thought loops, seizures, and heart failure in extreme cases.
- Alcohol - Combining alcohol with stimulants can be dangerous due to the risk of accidental over-intoxication. Stimulants mask alcohol's depressant effects, which is what most people use to assess their degree of intoxication. Once the stimulant wears off, the depressant effects will be left unopposed, which can result in blackouts and severe respiratory depression. If mixing, the user should strictly limit themselves to only drinking a certain amount of alcohol per hour.
- DXM - Combinations with DXM should be avoided due to its inhibiting effects on serotonin and norepinephrine reuptake. There is an increased risk of panic attacks and hypertensive crisis, or serotonin syndrome with serotonin releasers (MDMA, methylone, mephedrone, etc.). Monitor blood pressure carefully and avoid strenuous physical activity.
- MDMA - Any neurotoxic effects of MDMA are likely to be increased when other stimulants are present. There is also a risk of excessive blood pressure and heart strain (cardiotoxicity).
- MXE - Some reports suggest combinations with MXE may dangerously increase blood pressure and increase the risk of mania and psychosis.
- Dissociatives - Both classes carry a risk of delusions, mania and psychosis, and these risk may be multiplied when combined.
- Stimulants - 6-APDB may be dangerous to combine with other stimulants like cocaine as they can increase one's heart rate and blood pressure to dangerous levels.
- Tramadol - Tramadol is known to lower the seizure threshold[9] and combinations with stimulants may further increase this risk.
- MAOIs - This combination may increase the amount of neurotransmitters such as dopamine to dangerous or even fatal levels. Examples include syrian rue, banisteriopsis caapi, and some antidepressants.[10]
Serotonin syndrome risk
Combinations with the following substances can cause dangerously high serotonin levels. Serotonin syndrome requires immediate medical attention and can be fatal if left untreated.
- MAOIs - Such as banisteriopsis caapi, syrian rue, phenelzine, selegiline, and moclobemide.[11]
- Serotonin releasers - Such as MDMA, 4-FA, methamphetamine, methylone and αMT.
- SSRIs - Such as citalopram and sertraline
- SNRIs - Such as tramadol and venlafaxine
- 5-HTP
There is an increased risk of serotonin syndrome when 6-APDB is taken with many antidepressants, particularly selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs). Additionally, if 6-APDB is taken with SSRIs and SNRIs, the 6-APDB will be significantly less powerful or may have no distinguishable effects at all.
Legal status
- Australia and New Zealand: Certain countries contain a "substantially similar" catch-all clause in their drug law, such as New Zealand and Australia. This includes 6-APDB as it is similar in chemical structure to the class A drug MDA, meaning 6-APB may be viewed as a controlled substance analogue in these jurisdictions.[citation needed]
- Canada: 6-APDB is Schedule III in Canada as it is an analogue of MDA. The CDSA was updated as a result of the Safe Streets Act changing amphetamines from Schedule 3 to Schedule 1.[citation needed]
- France: 6-APDB is classified as a narcotic since May 9, 2018, alongside other substances derived from benzofuran.[12]
- Germany: 6-APDB is controlled under the NpSG (New Psychoactive Substances Act)[13] as of November 26, 2016.[14] Production and import with the aim to place it on the market, administration to another person and trading is punishable. Possession is illegal but not penalized.[15]
- Italy: 6-APDB is a prohibited substance in Italy.[16]
- Sweden: 6-APDB is prohibited in Sweden as a "health hazard" as of 2009.[citation needed]
- Switzerland: 6-APDB is a controlled substance specifically named under Verzeichnis E.[17]
- United Kingdom: On June 10, 2013, 6-APDB and a number of analogues were classified as Temporary Class Drugs in the U.K. following an ACMD recommendation. On March 5, 2014, the U.K. Home Office announced that 6-APDB would be made a class B drug on 10 June 2014 alongside every other benzofuran entactogen and many structurally related drugs.[18]
- United States: 6-APDB is unscheduled in the United States. It may be considered an analog of MDA (which is a Schedule I drug under the Controlled Substances Act). As such, the sale and possession for the purposes of human consumption or could be prosecuted as crimes under the Federal Analog Act.[citation needed]
See also
External links
References
- ↑ Monte, A. P., Marona-Lewicka, D., Cozzi, N. V., Nichols, D. E. (12 November 1993). "Synthesis and pharmacological examination of benzofuran, indan, and tetralin analogues of 3,4-(methylenedioxy)amphetamine". Journal of Medicinal Chemistry. 36 (23): 3700–3706. doi:10.1021/jm00075a027. ISSN 0022-2623.
- ↑ Iversen, L., Gibbons, S., Treble, R., Setola, V., Huang, X.-P., Roth, B. L. (30 January 2013). "Neurochemical Profiles of some novel psychoactive substances". European journal of pharmacology. 700 (1–3): 147–151. doi:10.1016/j.ejphar.2012.12.006. ISSN 0014-2999.
- ↑ Fleckenstein, A. E., Volz, T. J., Riddle, E. L., Gibb, J. W., Hanson, G. R. (1 February 2007). "New Insights into the Mechanism of Action of Amphetamines". Annual Review of Pharmacology and Toxicology. 47 (1): 681–698. doi:10.1146/annurev.pharmtox.47.120505.105140. ISSN 0362-1642.
- ↑ 4.0 4.1 Elangbam, C. S. (October 2010). "Drug-induced Valvulopathy: An Update". Toxicologic Pathology. 38 (6): 837–848. doi:10.1177/0192623310378027. ISSN 0192-6233.
- ↑ 5.0 5.1 Droogmans, S., Cosyns, B., D’haenen, H., Creeten, E., Weytjens, C., Franken, P. R., Scott, B., Schoors, D., Kemdem, A., Close, L., Vandenbossche, J.-L., Bechet, S., Van Camp, G. (1 November 2007). "Possible association between 3,4-methylenedioxymethamphetamine abuse and valvular heart disease". The American Journal of Cardiology. 100 (9): 1442–1445. doi:10.1016/j.amjcard.2007.06.045. ISSN 0002-9149.
- ↑ Nimmo, S. M., Kennedy, B. W., Tullett, W. M., Blyth, A. S., Dougall, J. R. (October 1993). "Drug-induced hyperthermia". Anaesthesia. 48 (10): 892–895. doi:10.1111/j.1365-2044.1993.tb07423.x. ISSN 0003-2409.
- ↑ Malberg, J. E., Seiden, L. S. (1 July 1998). "Small changes in ambient temperature cause large changes in 3,4-methylenedioxymethamphetamine (MDMA)-induced serotonin neurotoxicity and core body temperature in the rat". The Journal of Neuroscience: The Official Journal of the Society for Neuroscience. 18 (13): 5086–5094. ISSN 0270-6474.
- ↑ Wolff, K., Tsapakis, E. M., Winstock, A. R., Hartley, D., Holt, D., Forsling, M. L., Aitchison, K. J. (May 2006). "Vasopressin and oxytocin secretion in response to the consumption of ecstasy in a clubbing population". Journal of Psychopharmacology. 20 (3): 400–410. doi:10.1177/0269881106061514. ISSN 0269-8811.
- ↑ Talaie, H.; Panahandeh, R.; Fayaznouri, M. R.; Asadi, Z.; Abdollahi, M. (2009). "Dose-independent occurrence of seizure with tramadol". Journal of Medical Toxicology. 5 (2): 63–67. doi:10.1007/BF03161089. eISSN 1937-6995. ISSN 1556-9039. OCLC 163567183.
- ↑ Gillman, P. K. (2005). "Monoamine oxidase inhibitors, opioid analgesics and serotonin toxicity". British Journal of Anaesthesia. 95 (4): 434–441. doi:10.1093/bja/aei210
 . eISSN 1471-6771. ISSN 0007-0912. OCLC 01537271. PMID 16051647.
. eISSN 1471-6771. ISSN 0007-0912. OCLC 01537271. PMID 16051647.
- ↑ Gillman, P. K. (2005). "Monoamine oxidase inhibitors, opioid analgesics and serotonin toxicity". British Journal of Anaesthesia. 95 (4): 434–441. doi:10.1093/bja/aei210
 . eISSN 1471-6771. ISSN 0007-0912. OCLC 01537271. PMID 16051647.
. eISSN 1471-6771. ISSN 0007-0912. OCLC 01537271. PMID 16051647.
- ↑ "Article Annexe IV - Arrêté du 22 février 1990 fixant la liste des substances classées comme stupéfiants" (in French). Légifrance. Retrieved September 23, 2022.
- ↑ "Anlage NpSG" (in German). Bundesministerium der Justiz und für Verbraucherschutz. Retrieved December 18, 2019.
- ↑ "Gesetz zur Bekämpfung der Verbreitung neuer psychoaktiver Stoffe" (PDF) (in German). Bundesanzeiger Verlag. Retrieved December 18, 2019.
- ↑ "§ 4 NpSG" (in German). Bundesministerium der Justiz und für Verbraucherschutz. Retrieved December 18, 2019.
- ↑ http://www.salute.gov.it/imgs/C_17_pagineAree_3729_listaFile_itemName_0_file.pdf
- ↑ "Verordnung des EDI über die Verzeichnisse der Betäubungsmittel, psychotropen Stoffe, Vorläuferstoffe und Hilfschemikalien" (in German). Bundeskanzlei [Federal Chancellery of Switzerland]. Retrieved January 1, 2020.
- ↑ The Misuse of Drugs Act 1971 (Ketamine etc.) (Amendment) Order 2014